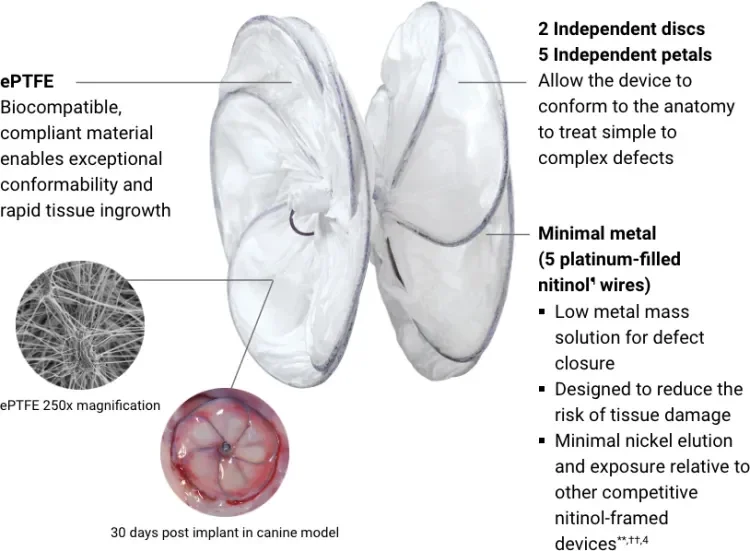
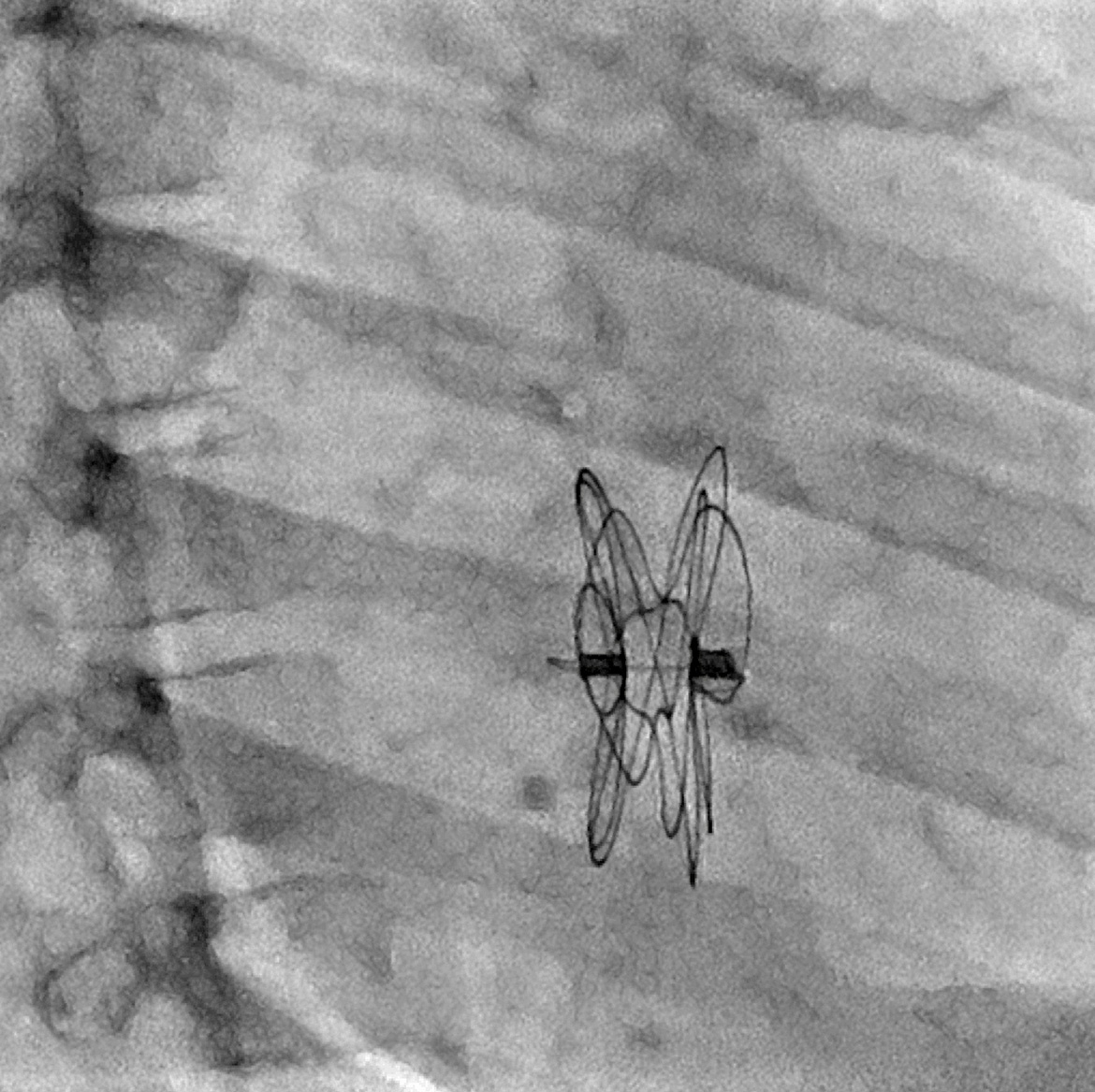
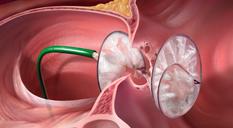
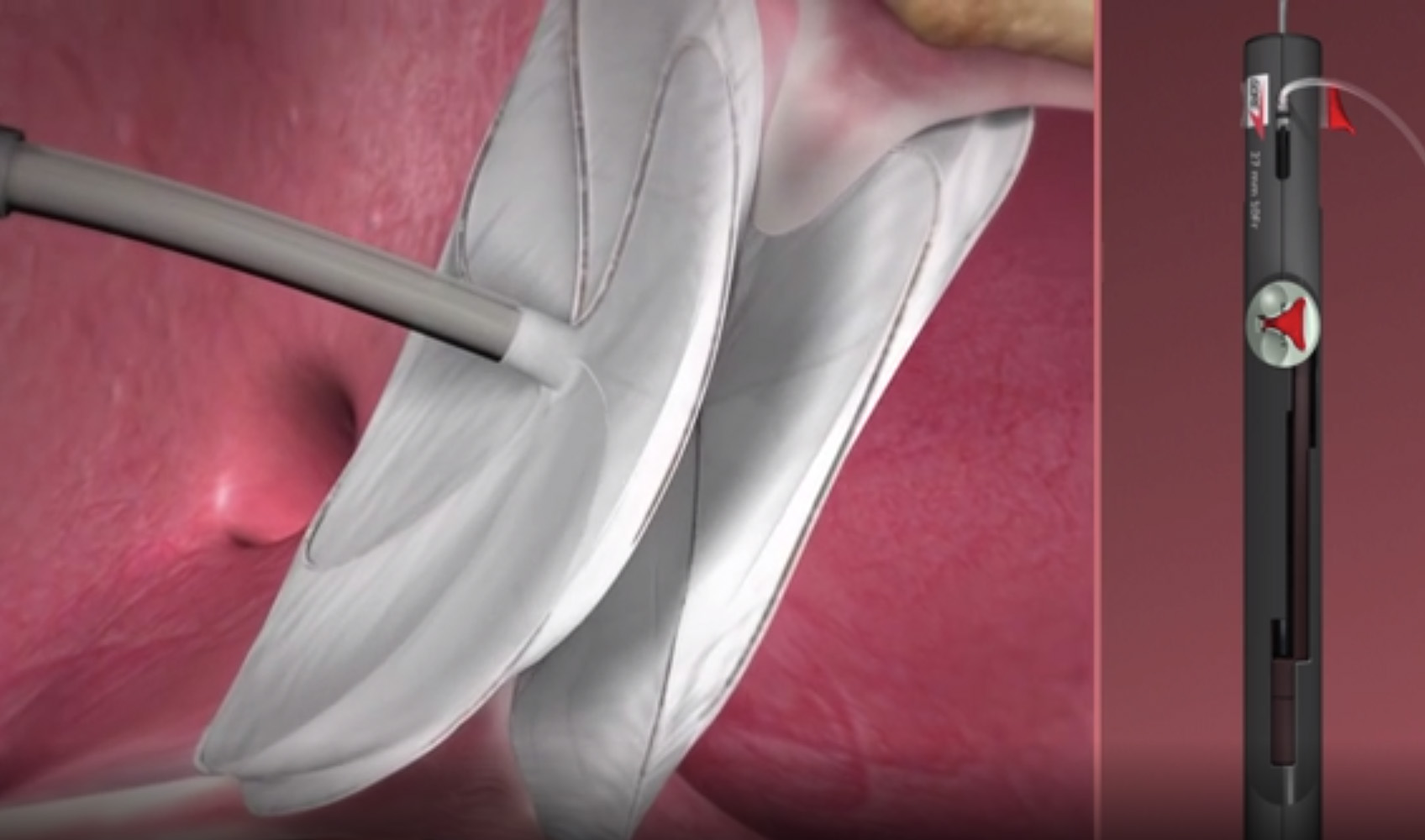
GORE CARDIOFORM est approuvé par la FDA pour le traitement des malformations de la muqueuse septale auriculaire - 555 Paper Mill Rd, Newark, DE 19711, USA

Trans-Septal Puncture Through Gore® Cardioform Septal Occluder Device - Step by Step Approach - ScienceDirect
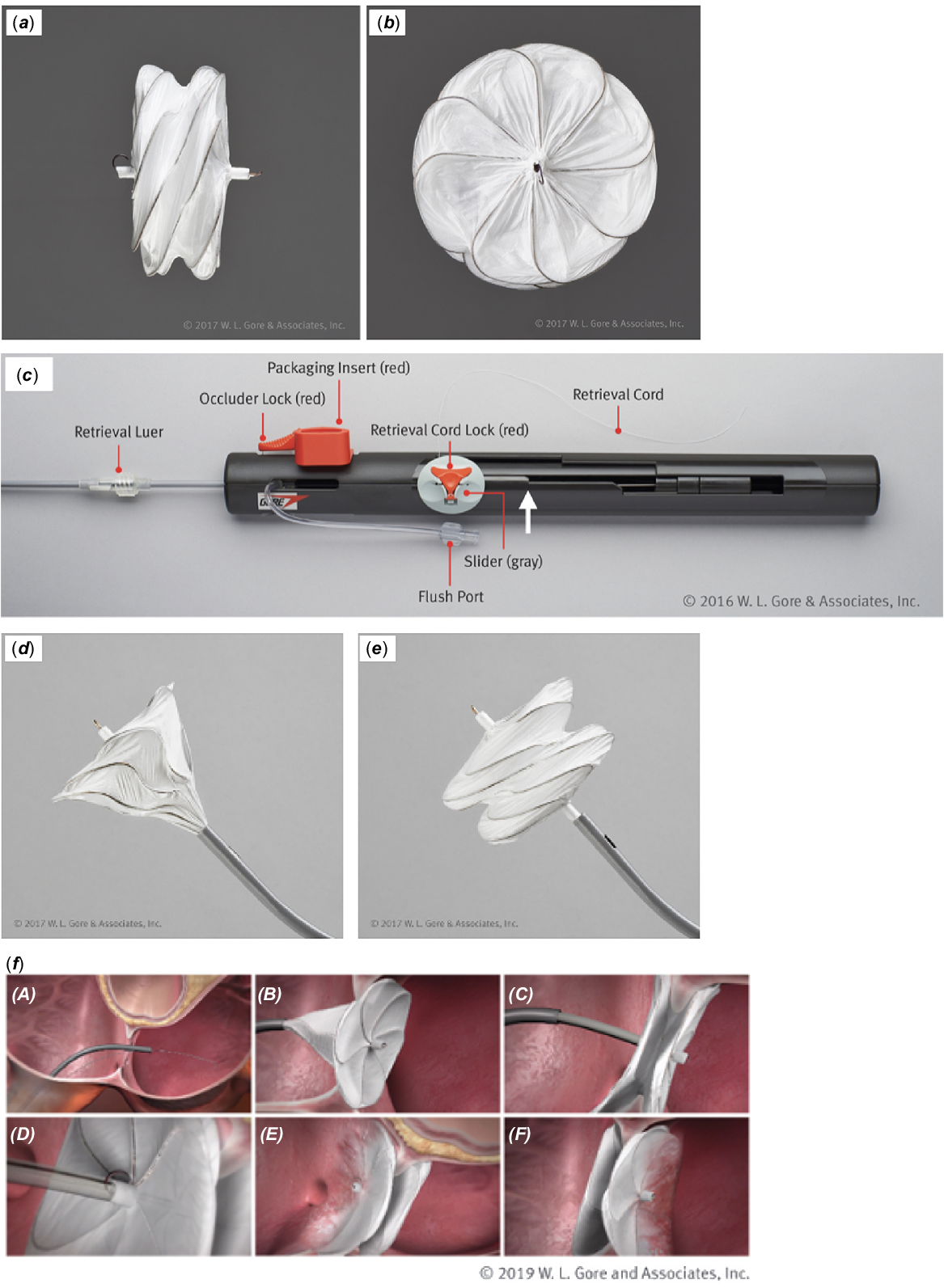
Gore Cardioform atrial septal occluder: deployment procedure and techniques for closing challenging secundum atrial septal defects | Cardiology in the Young | Cambridge Core

Mullins-Sheath Facilitated Delivery of Gore Cardioform ASD Occluder Devices for Closure of Large or Challenging Secundum Atrial Septal Defects

FDA Approves Gore Cardioform Septal Occluder for PFO Closure Prevent Recurrent Ischemic Stroke | DAIC

WCSA - Daily Highlighs - April 12, 2018 - GORE CARDIOFORM Septal Occluder Approved By FDA For PFO Closure - WCSA.WORLD

FDA Approves Gore's CARDIOFORM ASD Occluder To Treat Atrial Septal Defects | Medical Product Outsourcing