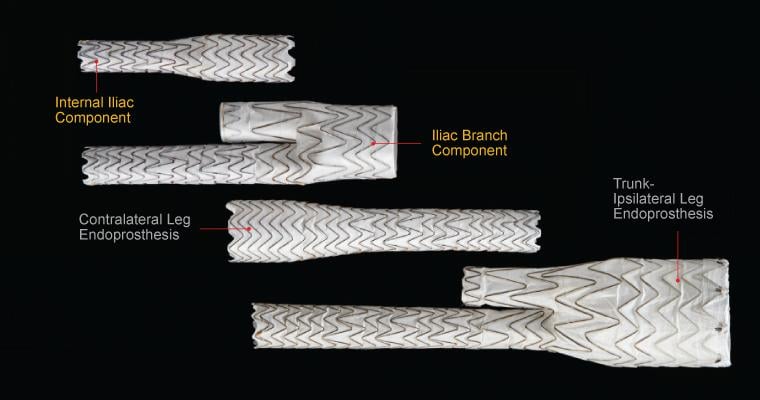
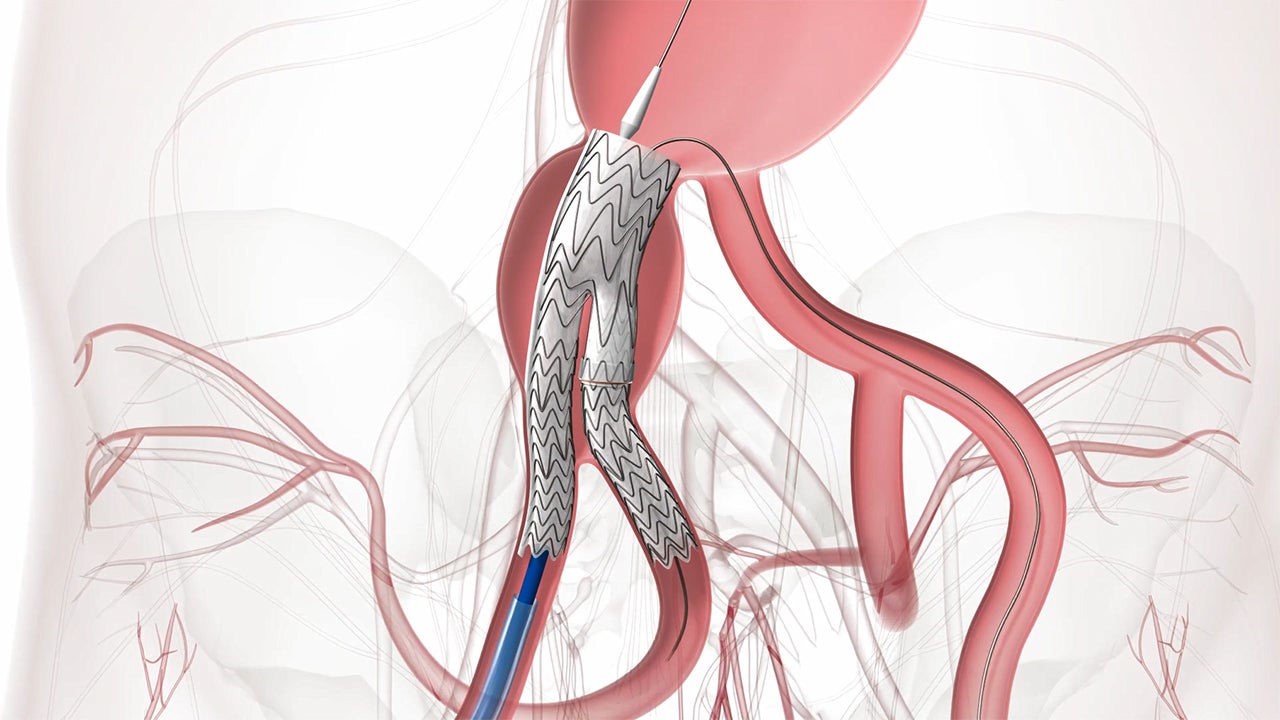
Techniques of Endovascular Aortoiliac Repair Using an Iliac Branch Endoprosthesis - Endovascular Today

Early results of expanding the anatomical indications for using a Gore Iliac branch endoprosthesis to treat aortoiliac and iliac aneurysms | Surgery Today

Outcomes of Gore iliac branch endoprosthesis with internal iliac component versus Gore Viabahn VBX - ScienceDirect
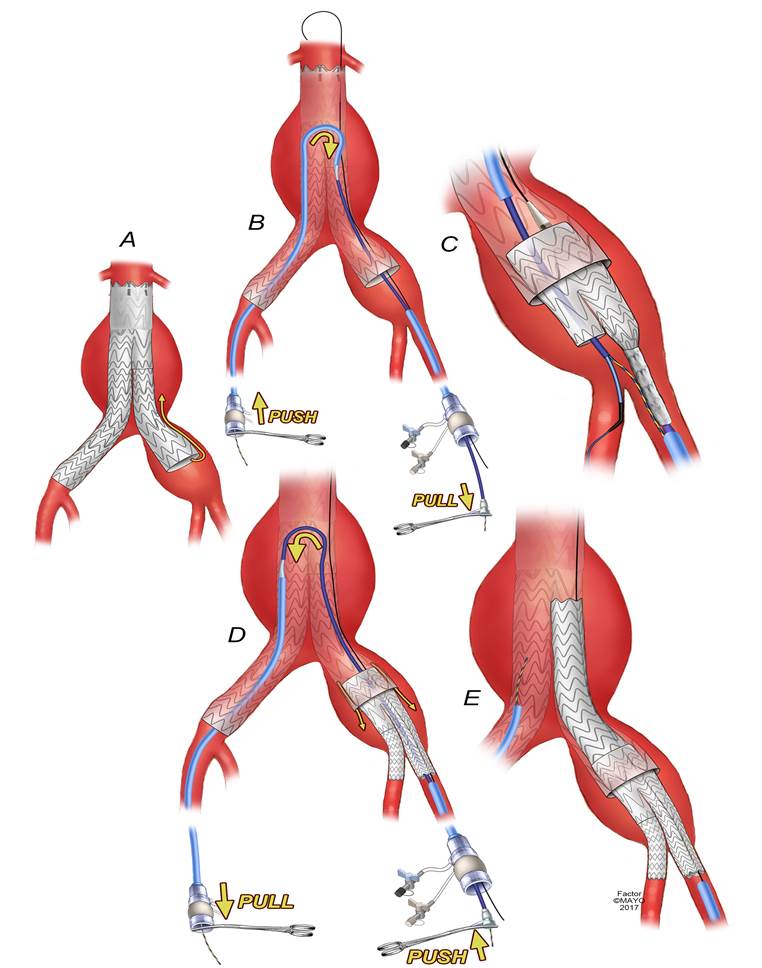
SCVS - Evaluation of a novel up-and-over transfemoral approach for endovascular repair using iliac branch endoprosthesis in patients with prior bifurcated endografts
Preliminary Experience with the GORE® EXCLUDER® Iliac Branch Endoprosthesis for Common Iliac Aneurysm Endovascul

Gore Iliac Branch Endoprosthesis for treatment of bilateral common iliac artery aneurysms - ScienceDirect

Recrutement du premier patient dans l'étude expérimentale sur l'endoprothèse AAA conformable GORE® EXCLUDER® avec système ACTIVE CONTROL | Business Wire
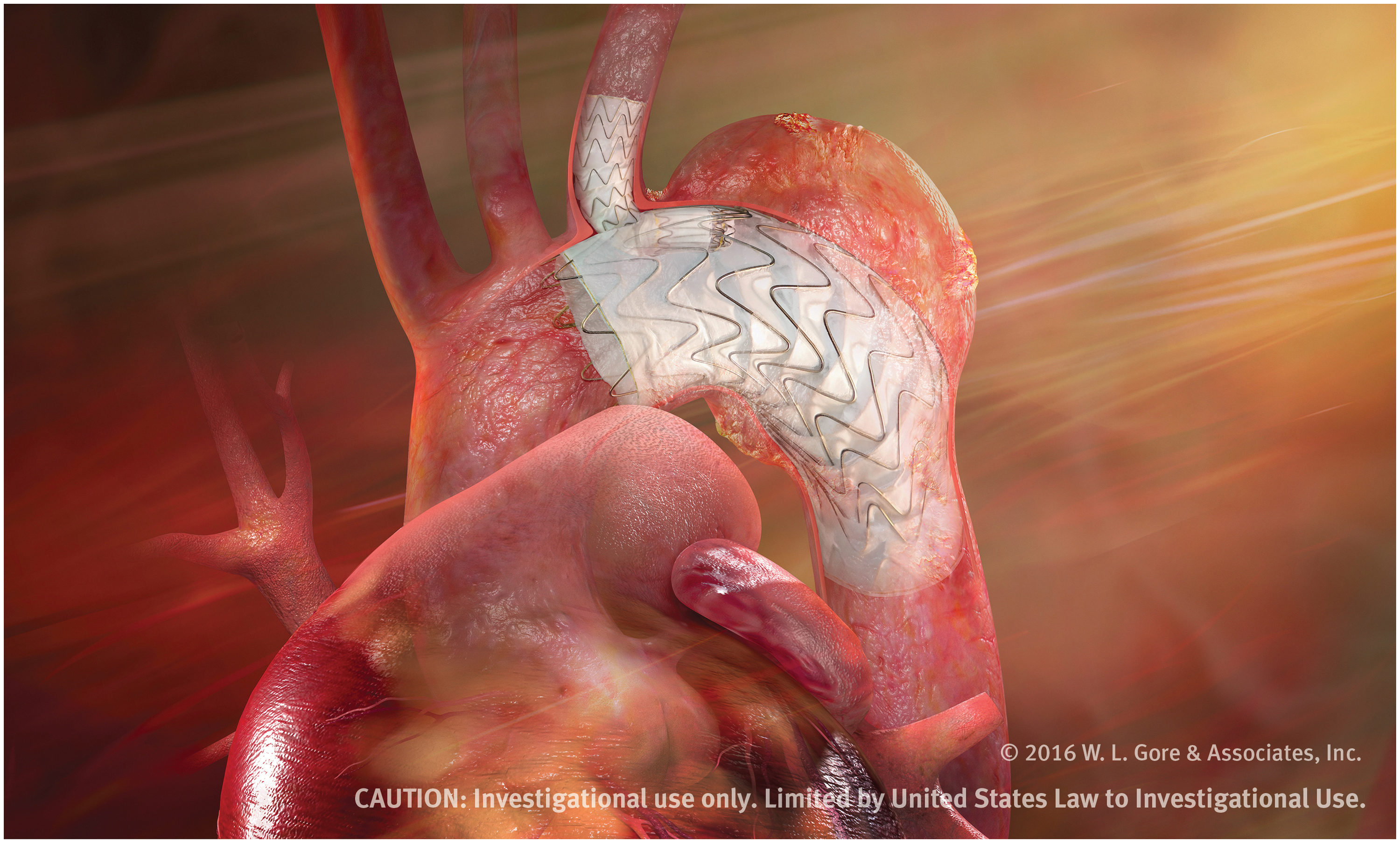
Le premier implant a été réalisé dans une étude pivot portant sur l'endoprothèse de branche thoracique GORE® TAG® | Business Wire

Gore Excluder IBE 'effectively prevented common iliac artery aneurysm enlargement' - Vascular Specialist